Logistics Technologist
Dublin, Leinster Job ID R0178245 Category Manufacturing & Supply Subcategory Manufacturing & Supply Business Unit Global Manufacturing & Supply Job Type Full timeBy clicking the “Apply” button, I understand that my employment application process with Takeda will commence and that the information I provide in my application will be processed in line with Takeda’s Privacy Notice and Terms of Use. I further attest that all information I submit in my employment application is true to the best of my knowledge.
Job Description
Logistic Technologist
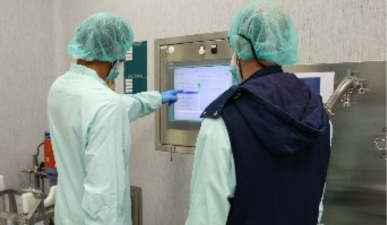
As our Logistics Technologist, you play a critical role because the position sits at the core of a regulated pharmaceutical supply chain, where accuracy directly impacts product quality and safety. The role combines hands‑on logistics activities with strict compliance requirements and continuous improvement. It also provides broad responsibility across operations, inventory, and procurement, making it a key operational function.
How will you contribute:
- Ensure smooth material flow including goods receipt, sampling, tanker offloading, material staging, storage, and shipment
- Execute logistics operations in compliance with cGMP, GWP, SOPs, and HSE regulations
- Operate and maintain forklifts, VNA trucks, PPTs, and other handling equipment
- Maintain accurate inventory through RFT receipts, timely stock transactions, cycle counts, and monthly reporting
- Support waste handling processes (solid and liquid) and contribute to process and efficiency improvements
- Assist procurement activities including stock monitoring, purchase order creation, supplier communication, and system updates
- Complete all required documentation and report deviations or abnormal events promptly
What you bring to Takeda:
- Leaving Certificate required; further education in Supply Chain or Material Handling is an advantage
- 2–3 years of experience in a pharmaceutical or regulated environment; warehouse experience essential
- Valid forklift, VNA, and PPT licenses
- Experience with SAP or similar Warehouse Management Systems
- Strong communication skills, teamwork, flexibility, and ability to work independently in a regulated environment
- Proficient in Microsoft Office (Outlook, Excel, Word); ability to work at heights
- Dangerous goods handling training, Lean/Six Sigma experience, First Aid/ERT training
What you can look forward to with us:
Competitive Salary: Including performance-based bonuses.
Retirement Plan: Employer contributions to your retirement plan.
Comprehensive Insurance: Employer-funded private medical insurance with dependants’ cover, life insurance, and flexible options like dental insurance, partner life insurance, and serious illness protection.
Electric Charging Points: Available at parking locations.
Employee Assistance Program: Support for personal and professional challenges.
Wellbeing and Engagement: Dedicated teams to support your wellbeing.
Family-Friendly Policies: Supportive policies for a balanced work-life.
Generous Vacation: 26 vacation days plus additional days for service milestones and humanitarian volunteering leave.
Development Opportunities: Coaching, mentoring, educational programs, and formal training.
Subsidized Canteen: Enjoy meals at a reduced cost.
Flexible Benefits: Options like fuel card, bike to work, commuter ticket, and insurance deals.
More about us:
At Takeda, we are transforming patient care through the development of novel specialty pharmaceuticals and best in class patient support programs. Takeda is a patient-focused company that will inspire and empower you to grow through life-changing work.
Certified as a Global Top Employer, Takeda offers stimulating careers, encourages innovation, and strives for excellence in everything we do. We foster an inclusive, collaborative workplace, in which our teams are united by an unwavering commitment to deliver Better Health and a Brighter Future to people around the world.
Empowering our people to shine:
Takeda is proud in its commitment to creating a diverse workforce and providing equal employment opportunities to all employees and applicants for employment without regard to race, color, religion, sex, sexual orientation, gender identity, gender expression, parental status, national origin, age, disability, citizenship status, genetic information or characteristics, marital status, or any other characteristic protected by law.
If you are interested in this opportunity, we look forward to receiving your application via our online tool!
Locations
Grange Castle, IrelandBase Salary Range:
€28,300.00 - €38,940.00For information about our benefits, please click here.